RNA viruses need to integrate control signals for genome replication, transcription of sub-genomic mRNAs and viral protein synthesis within the confined space of their genomes. These processes are often orchestrated by RNA structures, which are recognized by viral and host proteins and allow for efficient multiplication of the virus in the cell.
The RNA genome of SARS-CoV-2 harbors essential RNA structures at its 5′- and 3′-ends as well as in the frameshifting region (Fig. 1). Although the structures are highly conserved among betacoronaviruses, the function of most of them is unknown.
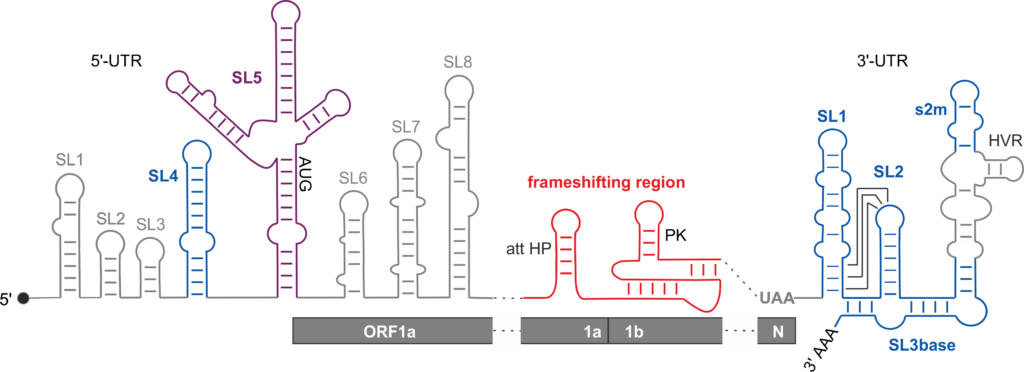
In collaboration, we analyzed the RNA folding of these functional SARS-CoV-2 RNA structures and develop small molecule ligands for RNA inhibition as antivirals. In current research, we investigate the structure-function-relationship of individual RNA folds and their mode of interaction with small molecules and proteins.
